 
Technical information, teaching suggestions, and related resources that complement this Concept Builder are provided on the Notes page. On their own, solutions such as K I ( a q ) (Potassium Iodide) and P b ( N O 3 ). For example, to determine if lead (Pb2+) is present in the. A precipitation reaction occurs when a solid forms from mixing two solutions. Learners and Instructors may also be interested in viewing the accompanying Notes page. Precipitation reactions are commonly used to identify if certain ions are present in a solution. Upon mixing the two, A g C l ( Silver chloride, a white compound) will instantly precipitate. As all of the other substances are soluble in water we can rewrite the equation. However, the 18 different groups of questions can be printed. 91 1 1 2 Add a comment 7 Answers Sorted by: 11 Heres one easy way : Make a solution of salt ( N a C l) in water, and another solution of Silver Nitrate ( A g N O X 3 ), which you can buy easily) in water. There is no need for an activity sheet for this Concept Builder. Users are encouraged to open the Concept Builder and explore. One of the best examples of precipitation reactions is the chemical reaction between potassium chloride and silver nitrate, in which solid silver chloride is. The built-in score-keeping makes this Concept Builder a perfect candidate for a classroom activity. All Group 1 ions form soluble salts, sodium nitrate, NaNO3(aq), is soluble. They are organized into 16 different Question Groups and spread across the three activities. Question-specific help is provided for each of the 16 situations. Molecular Equation sodium nitrate, NaNO3, is soluble. There are a total of 48 questions in this Concept Builder. In the third and final activity - Net Ionic Equations - learners are given the chemical formulas or the names of the two ionic compounds that are mixed as aqueous solutions and must identify the net ionic equation for the precipitation reaction that results. In the second activity - What's Going Down? - learners identify the chemical formula of the precipitate (or insoluble compound) that forms when two aqueous solutions of an ionic compound are mixed. In the first activity - Soluble or Insoluble? - learners must identify three ionic compounds as being either soluble or insoluble based on their given name or chemical formula a list of Solubility Rules is provided. The Precipitation Reactions and Net Ionic Equations Concept Builder challenges learners to identify the chemical formula of the precipitate that is formed when two aqueous solutions of an ionic compound are mixed AND to identify the net ionic equation for such a precipitation reaction. The Concept Builder includes three scaffolded activities. Precipitation Reactions and Net Ionic Equations 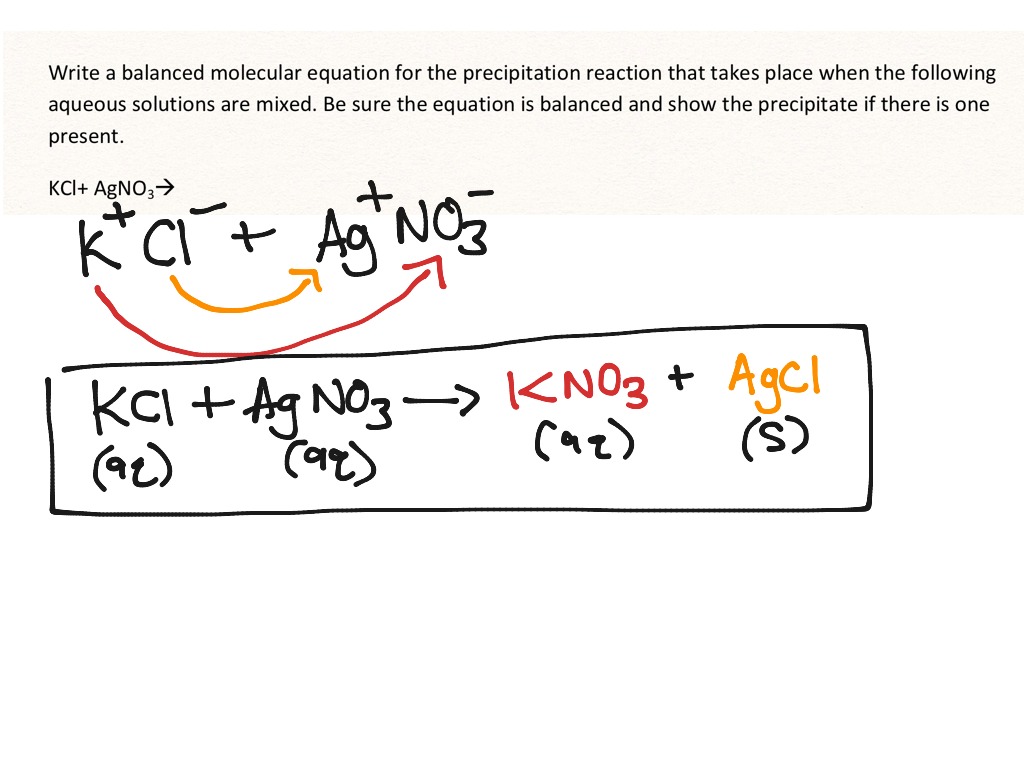
Concept Builders » Chemistry » Precipitation Reactions 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
